Gas, clay and girl power!
NEMESIS experiment can finally start at HADES after five years of preparation
How fast, how much and in what direction do dissolved gas particles move in Boom Clay? The NEMESIS experiment, which is part of the public-public partnership (PPP) between SCK CEN and NIRAS/ONDRAF and receives technical support from EURIDICE, is looking into it further. Unusual? Yes, because for the first time, the research is happening not on a lab scale, but on a large scale at the underground laboratory HADES. "Last Tuesday, the experiment began a course for at least five years. Fun fact: the scientific ‘triggers’ for the project are exclusively ladies. In our sector, you rarely come across that," says a proud Elke Jacops, project leader for the NEMESIS project (NEon diffusion in the MEgas In Situ).

Scaling up from centimetres to metres
Since 2009, SCK CEN has been measuring the diffusion coefficients of gases in clay at the above-ground labs, and there is now a large and reliable dataset available. Only so far, the measurements were made on samples only a few centimetres in size. To what extent are these values valid at large scales – for example, the Boom Clay at HADES? "To answer that question, we are conducting the NEMESIS experiment, where we want to measure the diffusion of neon gas over a distance of more than a metre and in different directions: horizontally and vertically," explains Elke Jacops.
Where does the gas come from and why are we watching it closely?
When we store some types of waste at an underground disposal site, they will begin to rust over time. While they rust, hydrogen gas is formed. This gas will move very slowly through the clay by diffusion. But when more gas is produced than what can spread through diffusion, gas bubbles will form and the pressure will rise. When the pressure gets too high, this can have a negative effect on the barrier function of the clay. To make an accurate estimate of the impact of this gas production, it is important to know how much gas is being created, as well as how much gas can spread back through diffusion. The speed at which the gas diffuses is called the diffusion coefficient.
What is diffusion?
Diffusion is the phenomenon whereby substances spread from a place of high concentration to where there is less of it. Imagine a room with a nice smell, like freshly baked cookies. If you open the door, the cookie smell will eventually spread throughout the house, even if you don't have the cookies with you. This is diffusion, whereby particles, such as the smell of cookies, naturally move and spread until they are evenly dispersed.
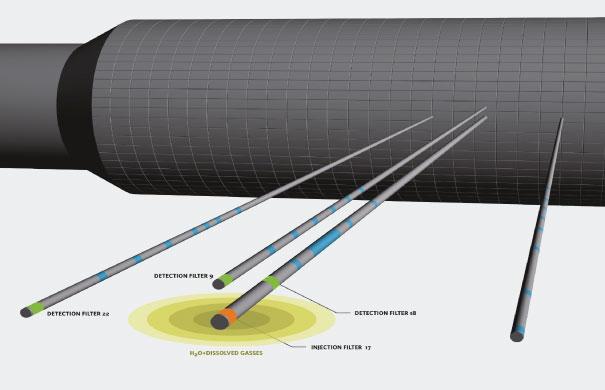
MEGAS & neon gas
Inert neon gas was chosen for this experiment because it is similar in size to hydrogen – which is the most relevant gas at a disposal site – and because neon does not naturally occur in the subsurface, which makes measurement easier. The team prepared the experiment for five years by modifying and transforming the setup formerly known as the MEGAS facility (installed in the early 1990s) into the NEMESIS facility. There, the neon is injected from an injection filter into the clay, after which it dissolves in water and diffuses through the clay.
Elke: "With three measurement filters placed at different distances, we can measure the quantity of neon transported by diffusion from the injection filter to these measurement filters in the clay. This is how we can study gas diffusion at a larger scale and under conditions more representative of a real depository, validating the gas diffusion coefficients from laboratory scale (cm) to a larger scale (metres)."

NEMESIS' leading ladies
The research combines the strengths of SCK CEN, EIG EURIDICE and NIRAS/ONDRAF. One fun fact is that this exploitation phase of the research is being supervised and monitored by four ladies: Elke Jacops, project leader at SCK CEN; Séverine Levasseur, scientific advisor at NIRAS/ONDRAF; Anneleen Vanleeuw as a technical laboratory technician at SCK CEN and Li Yu, scientifically responsible for transport calculations at SCK CEN. Elke emphasizes: "Of course, we always received and are still receiving support from many colleagues."
The team does not expect the initial results for another three years, but in the meantime, they are not sitting on their hands. "We're continuously monitoring the variation in pressure, temperature and water level. Based on the pressure drop we will be measuring in the first three months, we will already be able to make a very initial estimate of the diffusion coefficient. After three months, we'll also start our sampling to monitor the neon concentration on a regular basis and we're going to keep this up for at least three years," Elke concludes.
Also see the paper published in Geological Society, London, Special Publications.
Related articles
 23 April '24
23 April '24 29 February '24
29 February '24 13 February '24
13 February '24